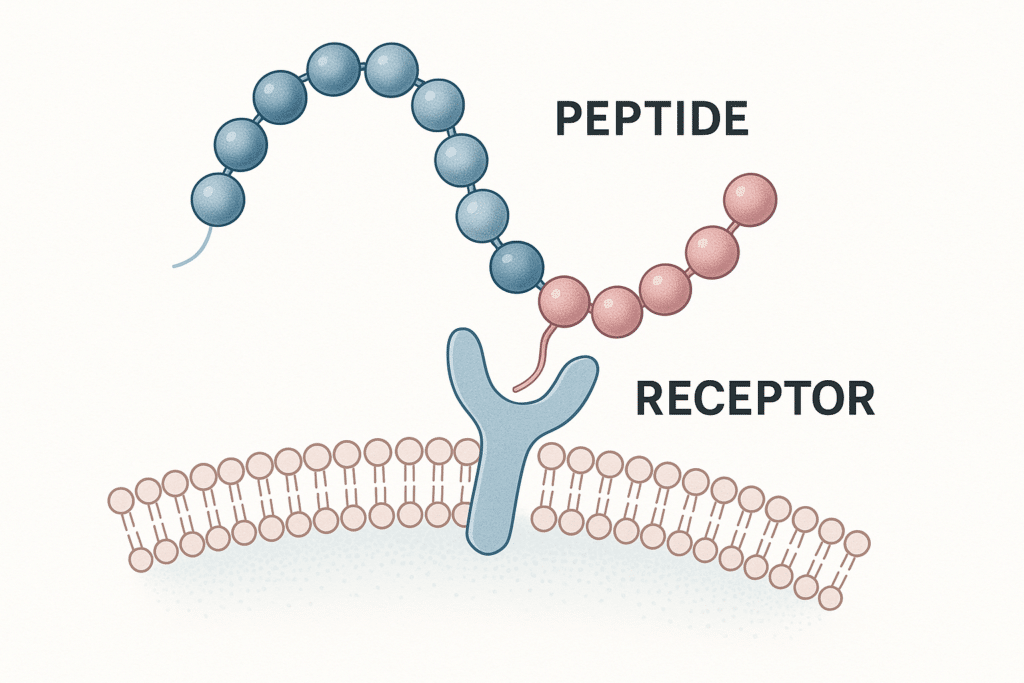
Quick summary: Repeated freeze–thaw cycles can introduce stress that may contribute to degradation, aggregation, or variability in biomolecules. For research peptide workflows, a simple preventive practice is aliquoting—splitting stock into single-use portions to reduce repeated thawing and refreezing. This guide is research-focused and does not provide dosing or administration instructions.
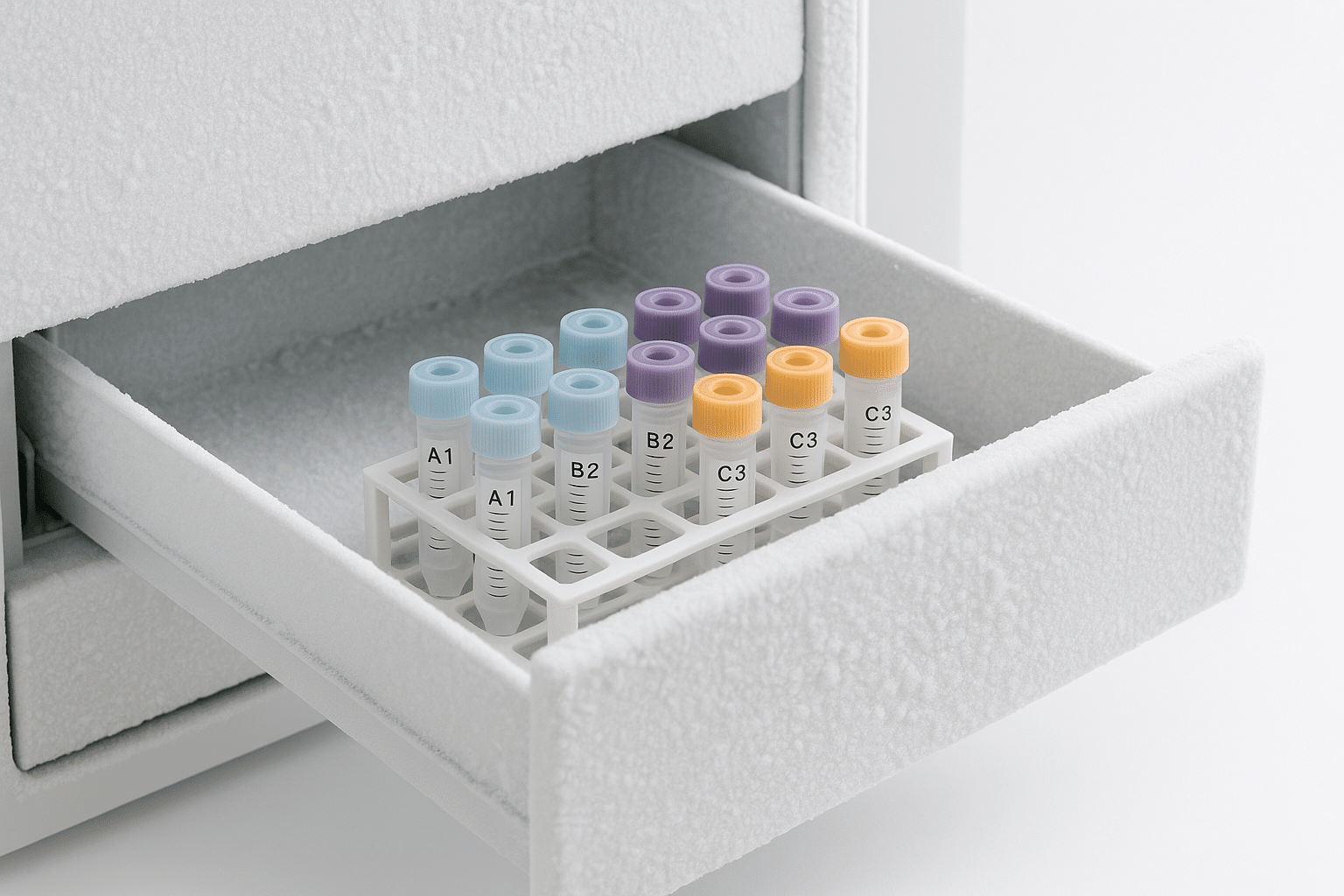
Table of contents
- What happens during freeze–thaw cycling?
- Why aliquoting helps (and when it matters most)
- A simple lab workflow to reduce freeze–thaw risk
- Common handling mistakes to avoid
- FAQ
- References
What happens during freeze–thaw cycling?
During freezing, water forms ice crystals and solutes can become more concentrated in the remaining liquid phase (often called a freeze-concentrated environment). During thawing, the system transitions again—potentially altering local concentrations, pH microenvironments, and interfaces. In many biomolecule contexts, these changes can contribute to structural stress and variability across cycles.
While peptide behavior depends on sequence and formulation, the general research handling principle is consistent: minimize unnecessary freeze–thaw exposure unless the material is specifically validated as stable through multiple cycles.
Why aliquoting helps (and when it matters most)
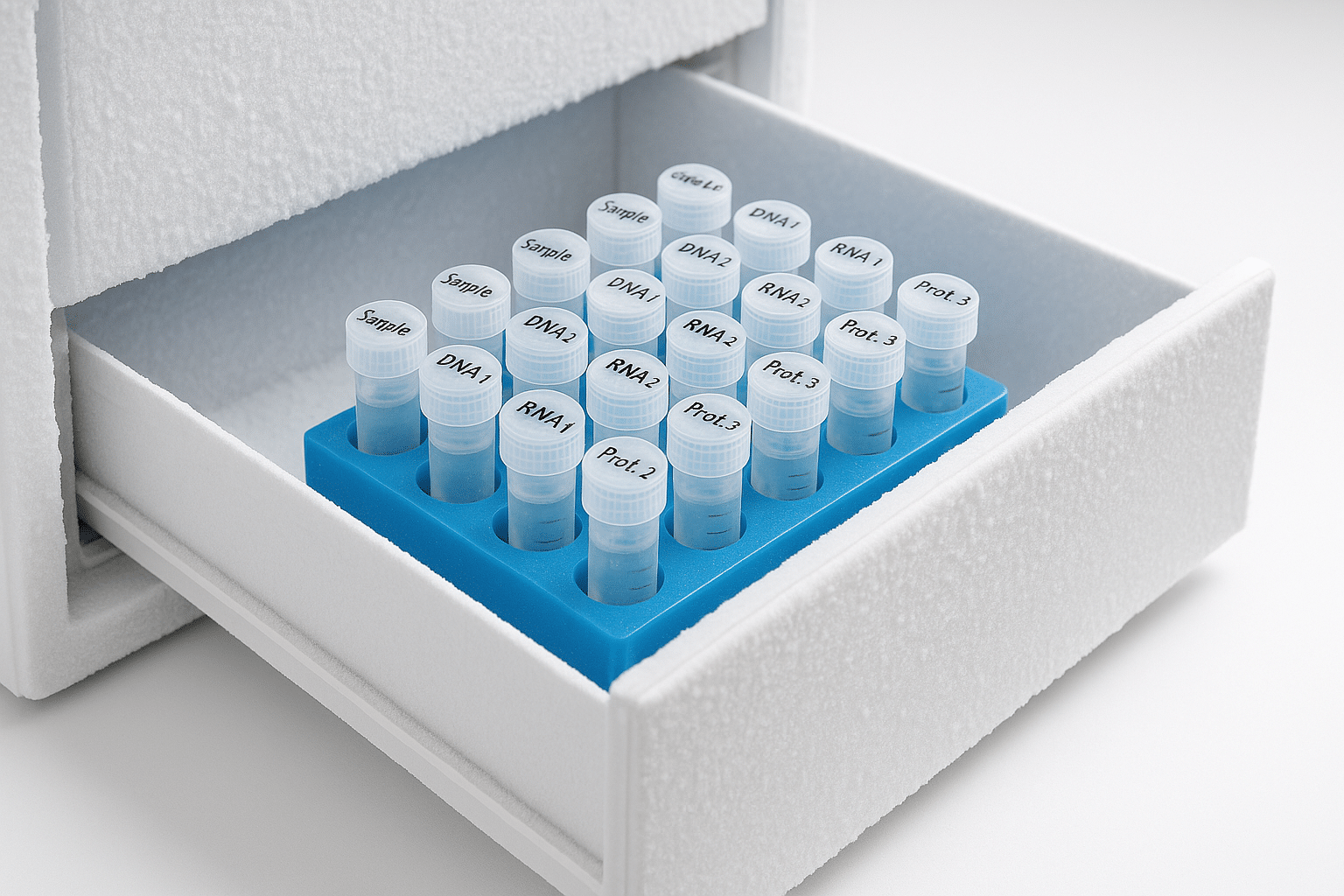
Aliquoting is simple: instead of storing one large vial that you thaw repeatedly, you split into smaller, single-use portions. This can help by:
- Reducing repeated temperature cycling on the same material
- Improving consistency between experiments (each aliquot is treated similarly)
- Lowering contamination risk (less repeated opening)
Aliquoting is especially useful when:
- You expect frequent use over weeks/months
- The peptide is stored in solution
- You’re running assays where small stability changes affect results
A simple lab workflow to reduce freeze–thaw risk
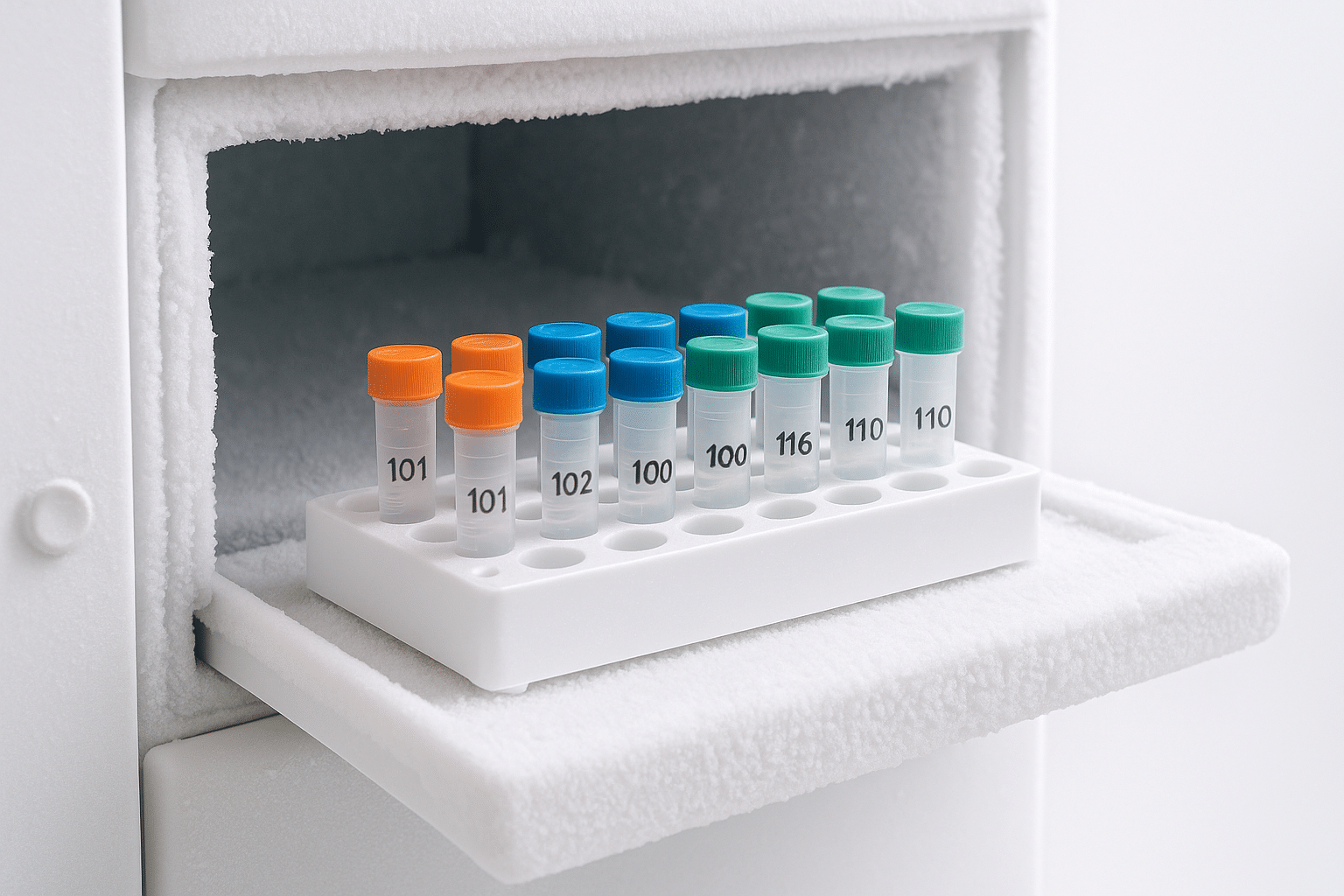
- Plan single-use volumes based on typical experiment needs.
- Label clearly: peptide name, concentration (if applicable), solvent (if applicable), prep date, and initials.
- Store consistently in a known location and keep an inventory log.
- Use one aliquot per session rather than returning partially-thawed material to long-term storage.
Common handling mistakes to avoid
- Repeated thaw/refreeze of the same vial without stability validation.
- Long bench time (unnecessary room temperature exposure).
- Poor labeling leading to unknown age/conditions of a sample.
- Moisture exposure for lyophilized material (opening cold vials in humid air).
FAQ
Do freeze–thaw cycles always degrade peptides?
Not always. Stability depends on the specific peptide, formulation, and handling conditions. In research workflows, it’s generally safer to assume repeated cycles can introduce variability unless stability is confirmed.
Is aliquoting necessary for lyophilized peptides?
Often, the bigger risk for lyophilized material is moisture exposure during repeated opening. Smaller vial sizes or planned handling steps can reduce exposure. Aliquoting is more commonly discussed for solutions, but workflow decisions should be peptide- and lab-specific.
How many freeze–thaw cycles are “too many”?
There is no universal number. If repeatability matters, minimize cycles. If repeated cycling is unavoidable, consider validating stability for your specific peptide and assay context.
References
- PubMed: 22860060
- NIH/NCBI PubMed Central: PMC10526705
- PubMed: 12673768
- NIH/NCBI PubMed Central: PMC7956273
Disclaimer: This article is for educational and research information only. It does not provide medical advice, diagnosis, treatment, dosing, cycles, or administration instructions. Research materials may have legal and regulatory restrictions depending on jurisdiction. Follow applicable laws and institutional policies.
Commercial disclosure: This site may reference research product listings for informational purposes.