Quick summary: Tirzepatide and semaglutide are peptide-based molecules studied for their activity on incretin-related receptors. At a high level, semaglutide is commonly described as a GLP‑1 receptor agonist, while tirzepatide is commonly described as a dual GIP/GLP‑1 receptor agonist. This article is written for research and educational purposes only and does not provide medical advice, dosing, or administration protocols.
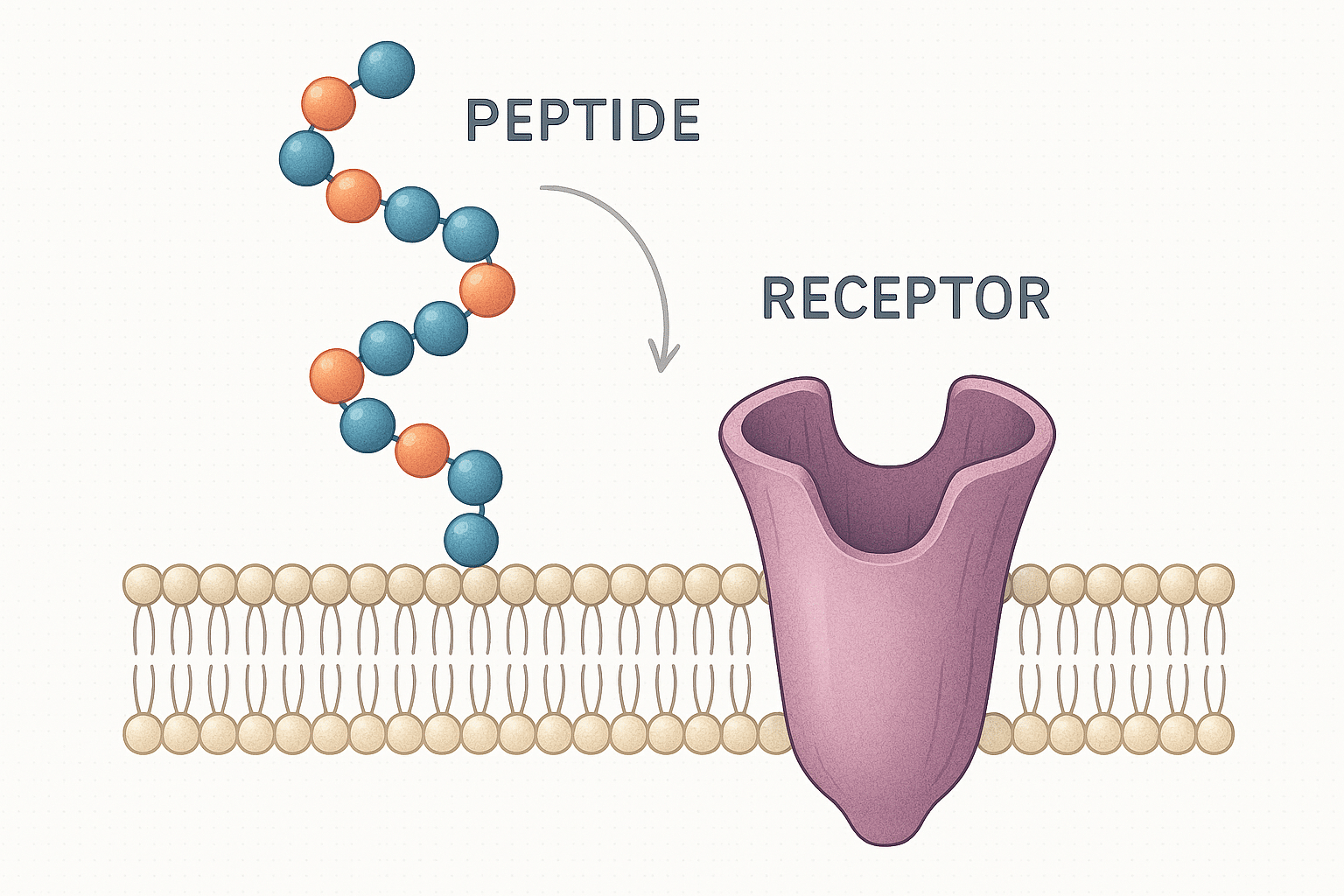
Table of contents
- High-level overview (what they are)
- Mechanisms: GLP‑1 vs dual GIP/GLP‑1 signaling
- Research landscape: what studies tend to evaluate
- Handling & storage considerations (research workflow)
- Research catalog listings (neutral links)
- FAQ
- References
High-level overview (what they are)
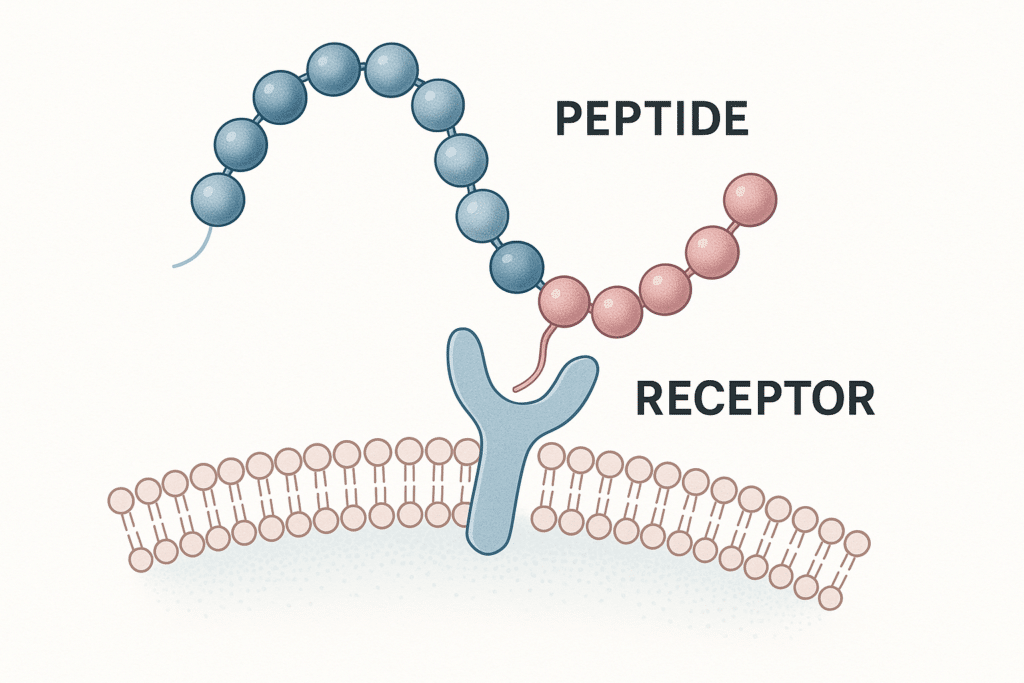
Both tirzepatide and semaglutide are peptide-based compounds discussed in the context of incretin receptor signaling. In research contexts, they are typically framed around receptor targets and downstream signaling effects rather than consumer outcomes.
Mechanisms: GLP‑1 vs dual GIP/GLP‑1 signaling
Semaglutide (GLP‑1 receptor agonism)
Semaglutide is widely described in the literature as a GLP‑1 receptor agonist. Research discussions often focus on receptor activation, downstream signaling pathways, and physiological effects in controlled study settings.
Tirzepatide (dual GIP/GLP‑1 receptor agonism)
Tirzepatide is commonly described as a dual agonist at GIP and GLP‑1 receptors (sometimes referred to as a “twincretin” in review literature). Research interest often centers on how dual-receptor activity may differ from single-receptor agonism in signaling and metabolic models.
Research landscape: what studies tend to evaluate
Depending on the model (in vitro, animal, or clinical research), studies may evaluate endpoints such as:
- Receptor binding and signaling (pathway activation, potency, bias)
- Pharmacokinetics / stability characteristics of peptide analogs
- Downstream metabolic markers in controlled settings
- Safety signals and adverse event profiles in clinical research
Important: Findings can be context-dependent. Mechanistic results in vitro do not automatically translate to real-world outcomes.
Handling & storage considerations (research workflow)
For peptides used in research workflows, common handling principles include minimizing moisture exposure (for lyophilized material), reducing repeated freeze–thaw cycles (often via aliquoting), and maintaining consistent labeling and storage conditions.
Research catalog listings (neutral links)
If you’re organizing a research catalog or comparing formats, here are neutral product listing links (no medical claims):
- Tirzepatide – 10mg (research listing)
- Tirzepatide – 30mg (research listing)
- Semaglutide – 10mg (research listing)
FAQ
Is tirzepatide the same as semaglutide?
No. They are commonly described with different receptor targeting profiles (GLP‑1 vs dual GIP/GLP‑1). In research contexts, that difference motivates comparative study design and interpretation.
Can research conclusions be generalized across peptides in the same class?
Not reliably. Small sequence and formulation differences can meaningfully affect stability, signaling, and outcomes in specific experimental contexts.
References
Disclaimer: This article is for educational and research information only. It does not provide medical advice, diagnosis, treatment, dosing, cycles, or administration instructions. Follow applicable laws and institutional policies.
Commercial disclosure: This site may reference research product listings for informational purposes.